Biomedical Resource Center
IACUC TRAINING
Purpose: Delineate required training for the IACUC (Institutional Animal Care and Use Committee), investigators who are listed on an IACUC approved protocol and/or personnel involved in the regular animal care or treatment of vertebrate animals and cephalopods covered under a protocol.
Rationale: The Animal Welfare Regulations (AWR) in Section 2.32 require the institution to ensure that all scientists, research technicians, animal technicians, and other personnel involved in animal care, treatment, and use are qualified and trained to perform their duties. Additionally, the institution must make training and instruction available in the specific areas outlined under 2.32(c). Please note: if you are working with biohazardous material or hazardous chemicals, additional training may be required.
Definitions: CITI: Collaborative Institutional Training Initiative, the online training platform which supplies training content
The IACUC requires that certain training must be completed before investigators can work with or provide care to vertebrate animals and cephalopods. This policy also includes personnel involved in regular animal care or treatment of vertebrate animals covered under an IACUC-approved protocol and BRC (Biomedical Resource Center) staff.
IACUC Members
IACUC Members:
Each IACUC member will be provided with a copy of the following:
The PHS (Public Health Service) Policy for the Humane Care and Use of Laboratory Animals.
The National Research Council (NRC): The Guide for the Care and Use of Laboratory Animals.
The ARENA/OLAW IACUC Guidebook.
The AVMA Guidelines on Euthanasia.
A copy of the OLAW (Office of Laboratory Animal Welfare) approved Assurance.
All new IACUC members are provided an orientation that covers the functions of an IACUC as well as an overview of the Animal Care and Use Program which provides training on methods for reporting concerns, humane practices of animal care and use (reduction, refinement, and replacement and methods to minimize pain and distress), use of hazardous agents when working with animals, zoonosis hazards, and what to do in the case of an injury. New members are mentored in protocol review and semi-annual review activities until they are comfortable conducting these activities on their own. In addition, members are required to take the “Essentials for IACUC Members” module offered through www.CITIProgram.org. The Chair and community members are also encouraged to complete CITI courses related to their roles. IACUC members are kept appraised of new policies and procedures via an IACUC dedicated SharePoint site.
Completion of one continuing education course is required annually and ongoing training on assorted topics is provided at a minimum of once a year during a designated IACUC meeting. Continued enrollment in the occupational health program is required.
Surgical Training
When working with rodents in research, maintaining proper aseptic techniques and providing appropriate surgical training are critical for ensuring animal welfare and research validity. The Institutional Animal Care and Use Committee (IACUC) plays a vital role in overseeing and ensuring that the procedures follow ethical guidelines, minimizing pain and distress for the animals.
Surgical Training for Rodents:
Surgical techniques must be learned and practiced to ensure the procedures are performed correctly and efficiently. Proper training and preparation are vital to prevent complications during surgery. All surgeons are required to complete hands on aseptic technique and surgical technique training with the attending veterinarian.
Training Elements:
- Surgical Skill Acquisition:
- Basic Procedures: Trainees often begin with non-invasive techniques and work their way up to more complex surgeries, such as implantation of devices or organ resection.
- Use of Animal Models: Often, training is performed using simulators, cadavers, or other models before conducting live surgeries on rodents.
- Anesthesia and Analgesia:
- Proper anesthesia is essential for rodent surgeries. Training includes understanding the correct dosing of anesthetics (e.g., isoflurane or injectable anesthetics like ketamine/xylazine), monitoring anesthesia depth, and ensuring post-operative analgesia is provided to minimize pain.
- Monitoring and Postoperative Care:
- Trainees should be familiar with how to monitor a rodent’s vital signs (e.g., temperature, respiratory rate, heart rate) during surgery and how to recognize signs of distress or complications.
- Postoperative care should include providing pain relief (e.g., opioids or NSAIDs), monitoring for infection, and ensuring proper recovery conditions.
- Ethics and IACUC Compliance:
- Surgeons must be familiar with the protocols approved by the IACUC, which include ensuring minimal discomfort, appropriate use of anesthetics, and adherence to the 3Rs principle (Replacement, Reduction, Refinement).
- The IACUC ensures that animal welfare standards are met, including appropriate surgical training and proper justification for any procedure.
- Hands-On Experience:
- Adequate hands-on experience under the guidance of an experienced surgeon is critical. Many institutions require trainees to complete a series of supervised surgeries and demonstrate competence before performing unsupervised surgeries.
IACUC Responsibilities in Surgical Training:
- Protocol Review: IACUC reviews all research protocols involving surgery to ensure that proper training, techniques, and post-operative care plans are in place.
- Oversight and Monitoring: IACUC is responsible for monitoring ongoing procedures to ensure that all aseptic practices and ethical standards are followed throughout the study.
- Training Documentation: IACUC may require documentation of training sessions, including certifications of successful completion and competency in the necessary surgical techniques.
Final Considerations:
- Recordkeeping: Maintain detailed records of training sessions, surgical procedures, and post-surgery monitoring. These records should include anesthesia protocols, surgical steps, and any complications that occurred.
- Refinement: Continue to refine surgical techniques over time to minimize harm to the animals. This includes adopting new technologies, improving pain management strategies, and using less invasive methods when possible.
In conclusion, aseptic techniques and proper surgical training for rodents are critical to maintaining high research standards and ensuring the humane treatment of animals. The IACUC’s role in overseeing these practices ensures that animal welfare is upheld while maintaining the integrity of the research.
Prior to training, please review: Principles for the New Surgeon
To schedule a surgicial training, please contact
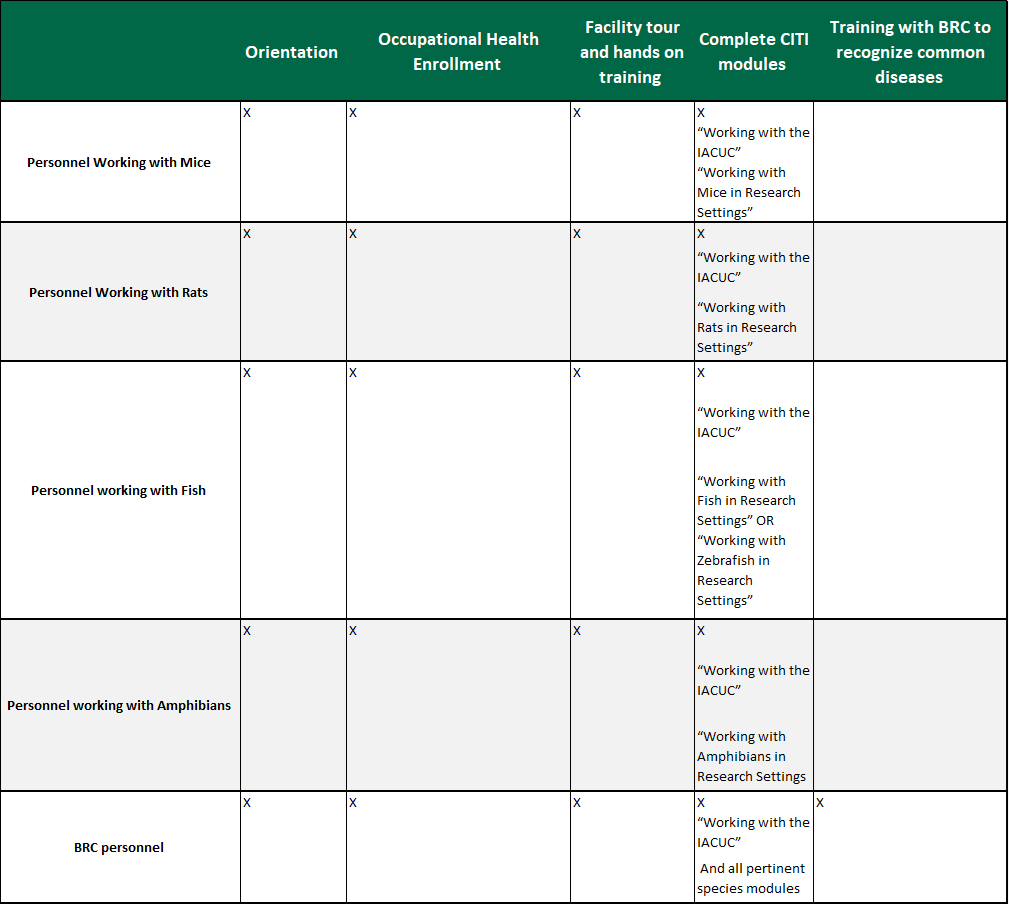
Scientists, Animal Technicians, and other Personnel involved with Animal Care
Scientists, Animal Technicians, and other Personnel involved with Animal Care:
(See Chart Below)
Training must be completed prior to working with animals and prior to protocol approval. All personnel are required to attend an orientation which provides training on methods for reporting concerns, humane practices of animal care and use, reduction, refinement, and replacement, as well as methods to minimize pain and distress, use of hazardous agents when working with animals, zoonosis hazards, and what to do in the case of an injury. A facility orientation is also required during which rodent users receive hands on training for animal handling and restraint. All personnel must be continually enrolled in the occupational health program.
All personnel and supervisors with roles in the care and use of animals at the Institution are to take online CITI training relevant to the species they will be working with as well as the CITI course entitled “Working with the IACUC.” Completion of one continuing education course is required annually. The course must be related to animal welfare or reduction, refinement, and replacement. This can be completed through webinars, CITI courses, AALAS (American Association for Laboratory Animal Science) courses, seminars, conferences, etc. The IACUC has delegated the determination of appropriate applicability of annual continuing education courses to the BRC staff.
BRC personnel must complete the above training and be trained on all BRC standard operating procedures, specific to the responsibilities within the animal care and use program. They are also trained to recognize common laboratory animal ailments.
Personnel performing surgery must complete a training session with the veterinarian on proper use of anesthetics, analgesics, and tranquilizers for the species studied as well as aseptic technique and suture training as applicable to the project.
Training in experimental methods such as specific animal manipulations and techniques will be conducted on an as needed based for the types of research being conducted and the species being studied.
Note: For investigators transferring from other facilities at which they have received similar training, verification of previous training may be accepted in lieu of some Institutional required training. Acceptance of previous training in lieu of the Institution’s training is solely at the IACUC’s discretion.
Zoonotic Agents of Concern
Zoonoses Associated with Rodents
This section provides information on various diseases that can be passed from mice, rats, gerbils, hamsters, guinea pigs and other rodent species to people. Often these diseases do not make the animal appear sick but can cause serious illness in humans. Persons with specific medical conditions such as a chronic illness, immunodeficiency and pregnancy may be at higher risk of developing disease or complications from a zoonotic disease and should consult with their physician before working with animals. Additional information on zoonotic diseases can be found on the Center for Disease Control and Prevention Website, Healthy Pets, Healthy People.
The majority of rodents housed at ACHE RIHWC are bred and raised under strict hygienic conditions and are free of pathogens that could be transferred to people. These rodents are called “specific pathogen-free” or “SPF” rodents. Rodents that are housed outdoors, captured from wild populations or that are purchased from a pet store or from a breeder who sells rodents as feed for reptiles may carry zoonotic diseases. If handling wild caught rodents, please refer to the Zoonoses fact sheet for wild rodents and the CDC hantavirus guidelines (CDC Hantavirus) for more information. Zoonotic diseases associated with rodents include rat bite fever, tularemia, hantavirus, lymphocytic choriomeningitis virus, leptospirosis, salmonellosis, campylobacterosis, and giardiasis.
Rat Bite Fever caused by Streptobacillus moniliformis or Spirillum minus is a bacterial infection of rodents that is transmitted through bites, scratches, direct contact with animals and their urine, saliva and feces or ingestion of contaminated food or water. Infected rodents typically exhibit no symptoms of disease. Tularemia is another bacterial infection of rodents. Infected rodents appear lethargic, but they may shed bacteria before showing illness. Tularemia is transmitted to people in the same manner as rat bite fever but in addition can be transmitted through the bite of an infected tick and via airborne transmission if feces, urine or body fluids are aerosolized. Both diseases in humans initially present as a fever, headache, swollen lymph nodes and possibly a rash or ulcer in the area of a recent bite or scratch wound. Any bites or scratches should be thoroughly washed immediately to minimize the chance of infection.
Rodents with hantavirus, lymphocytic choriomeningitis virus (LCMV) and leptospirosis usually do not exhibit signs of disease. The disease agents are typically shed in the urine of infected animals and people acquire the infection by inhalation, accidental ingestion and direct contact with contaminated urine or feces. These are occasionally transmitted from bite wounds and Leptospira can infect people through abraded skin. These diseases often initially appear as a mild flu-like illness in people but may progress to severe disease. LCMV infection can cause severe meningitis and hemorrhagic fever in people and is considered hazardous to the unborn fetus and may cause pre-term delivery or other complications in pregnant women. Please refer to the CDC Hantavirus guidelines (CDC Hantavirus) if working with wild rodents or in rodent-infested areas and buildings.
Salmonellosis, campylobacterosis, and giardiasis are acquired by contact and accidental ingestion of fecal material from infected rodents. Animals infected with these diseases may have diarrhea, but some may show no symptoms of disease. Any animal with diarrhea should be suspected of having a zoonotic disease. Symptoms in people include diarrhea, vomiting, and abdominal cramps.
_____________________________________________________________________________
Individuals with exposure to rodents and rodent housing environments may develop allergic reactions to animal proteins (allergens). Approximately 11-44% percent of individuals working with rats and mice will develop an allergic reaction to animal proteins and 5-10 percent of individuals will develop asthma. Personnel may be exposed to allergens through inhalation and contact with skin, eyes and mucous membranes. Animal allergens may be present in animal dander, hair, skin, urine, saliva, serum and any contaminated feed or bedding materials. Risk factors for developing an allergic reaction include history of previous allergies to animals. The signs and symptoms of an allergic reaction are nasal discharge and congestion, conjunctivitis, tearing and eye itching, skin redness, rash or hives and lower airway symptoms (coughing, wheezing and shortness of breath). Severe anaphylactic reactions to rodent bites have been reported. Individuals with symptoms suggestive of an allergic reaction related to a workplace allergen should report their concerns to their supervisor and consult a physician.
Wild, outdoor-housed and pet-store purchased rodents are much more likely to carry infections than those raised and housed in a laboratory setting. Transmission of zoonotic diseases from rodents is primarily by direct contact, bites, indirect contact with contaminated objects, oral ingestion or inhalation of aerosolized bedding, feces, and urine. We can protect ourselves from most diseases by using the following basic hygiene procedures:
- Do not eat, drink, apply cosmetics or use tobacco products while handling animals or in animal housing areas.
- Wear respiratory protection when appropriate.
- Handle animals safely to avoid bites and scratches. Thoroughly wash any bite or scratch wounds and report injuries.
- Wear gloves when handling animals, animal tissues, body fluids and waste and wash hands after contact.
- Wear dedicated protective clothing such as a lab coat or coveralls when handling rodents. Launder the soiled clothing separate from your personal clothes and preferably at the animal facility.
- Report ill animals so that they can receive veterinary care.
- Keep animal areas clean and disinfect equipment after using it on animals or in animal areas.
Most importantly, familiarize yourself about the animals that you will be working with and the potential zoonotic diseases associated with each species. If at any time, you suspect that you have acquired a zoonotic disease, inform your supervisor and seek medical care.
_____________________________________________________________________________
References
- Centers for Disease Control and Prevention. Healthy Pets, Healthy People. (Small Mammals | Healthy Pets, Healthy People | CDC)
- Centers for Disease Control and Prevention. Hantavirus Pulmonary Syndrome (HPS). (CDC – Hantavirus Pulmonary Syndrome (HPS) – Hantavirus)
